Research
Research Scope
The rapid emergence of multidrug resistant bacteria is a major global concern. A 2014 report by the UK government estimates that by 2050, antibiotic resistance will cause 300 million premature deaths, result in a loss of up to $100 trillion from the global economy and cause more premature deaths than cancer. Therefore, there is a pertinent need for new antimicrobial compounds and targets.
New Tools for New Targets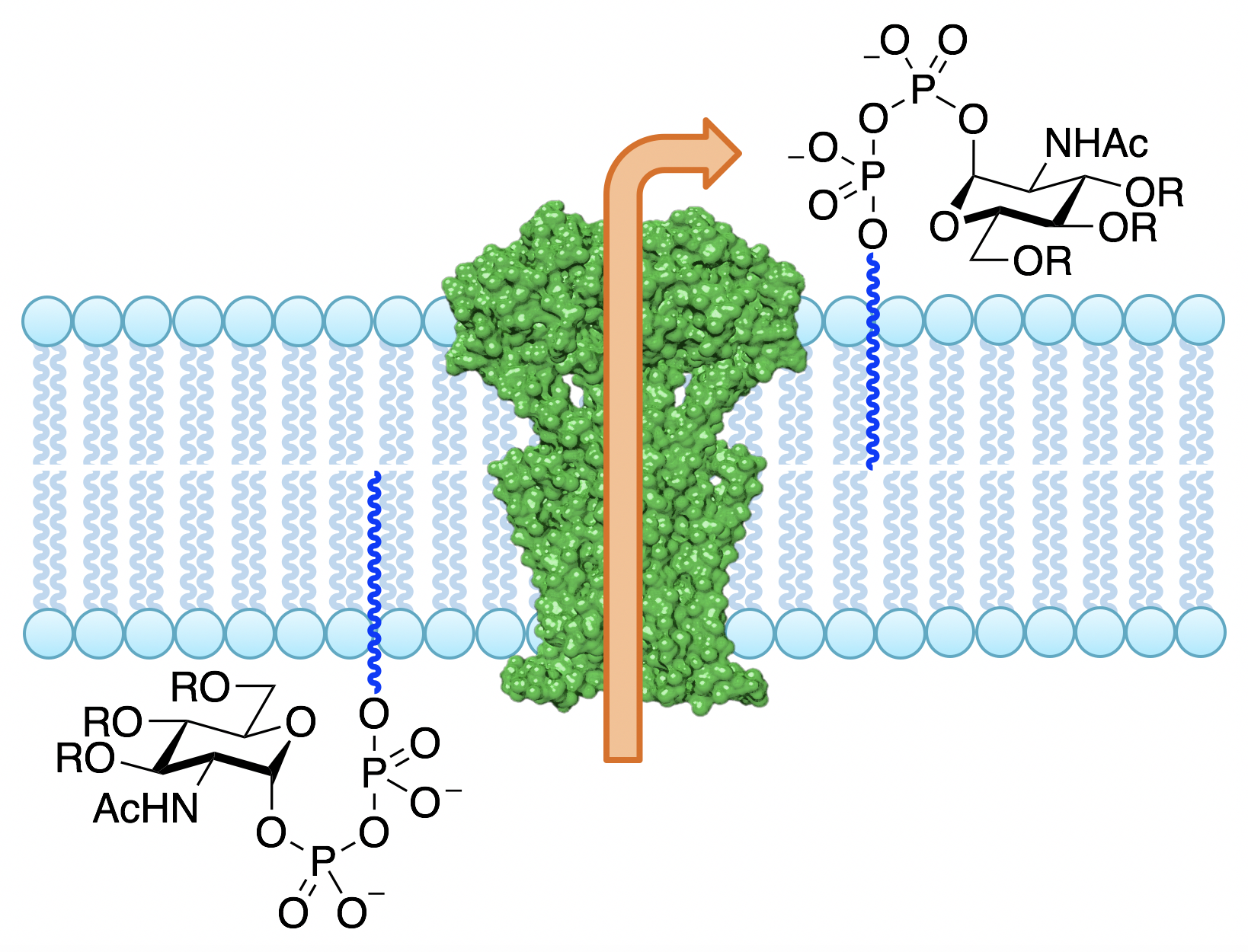
Bacteria produce several polysaccharides that are essential to their structural integrity, including peptidoglycan and lipopolysaccharide. As the enzymes and intermediates involved in their biosynthesis are unique to bacteria, they are excellent antibiotic targets. Our lab uses the tools of organic synthesis to prepare glycolipid substrates for probing the mechanism of action of these enzymes, as well as using structure-activity relationship studies to guide inhibitor design, with the goal of developing new antibiotic candidates.
New Antibiotics
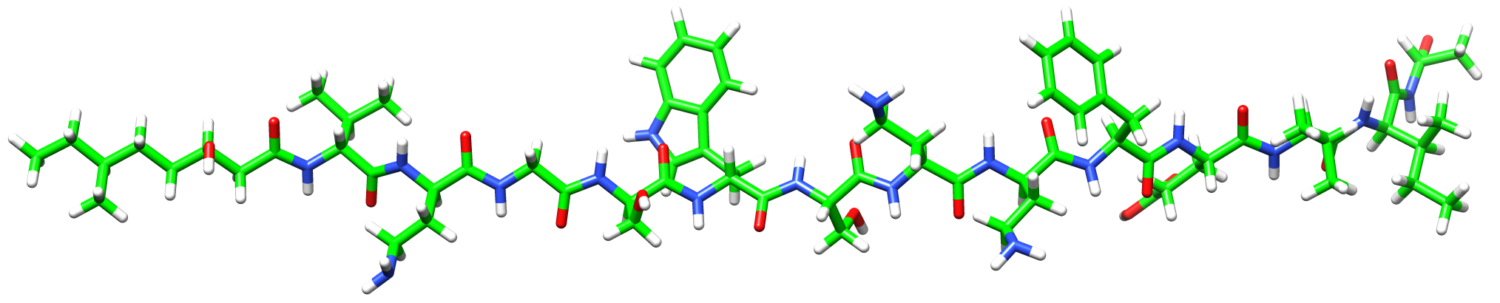
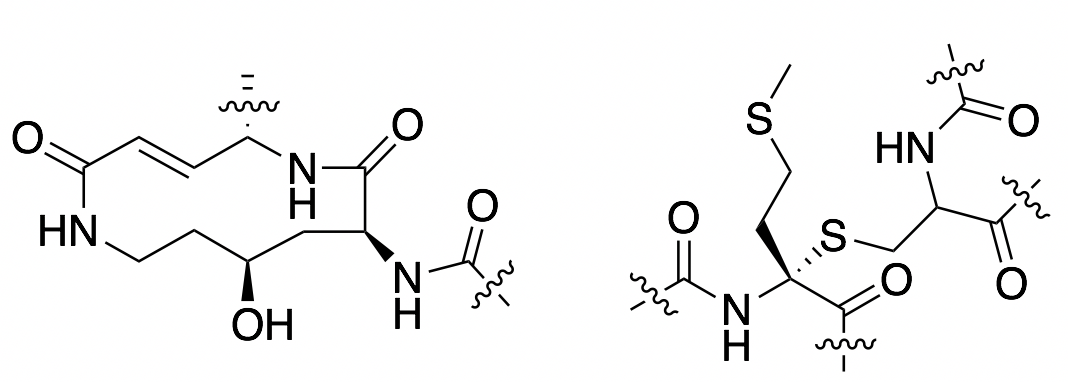 Antimicrobial peptides produced by bacteria are becoming increasingly important in the fight against multidrug resistance. These compounds typically target bacterial cell membranes, and as it is difficult for bacteria to reorganize their membranes, resistance development is often limited. Our lab develops new synthetic methods to access novel antimicrobial peptides, allowing their antimicrobial and/or anticancer activities to be explored. We also use molecular biology and bioinformatics to identify the mechanism by which they exert their therapeutic effect.
Antimicrobial peptides produced by bacteria are becoming increasingly important in the fight against multidrug resistance. These compounds typically target bacterial cell membranes, and as it is difficult for bacteria to reorganize their membranes, resistance development is often limited. Our lab develops new synthetic methods to access novel antimicrobial peptides, allowing their antimicrobial and/or anticancer activities to be explored. We also use molecular biology and bioinformatics to identify the mechanism by which they exert their therapeutic effect.